Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
To ensure a rapid path to implementation, IICC has established three active partnerships whereby members of the IICC Technical Team are assigned to specific partner organizations, becoming members of relevant work groups.Health Level Seven International (HL7) is the standards development organization (SDO) for IICC. Integrating the Healthcare Enterprise (IHE) is the integration profile developer for the IICC initiative. Clinical and Laboratory Standards Institute (CLSI) is the laboratory standards publisher for the IICC initiative.
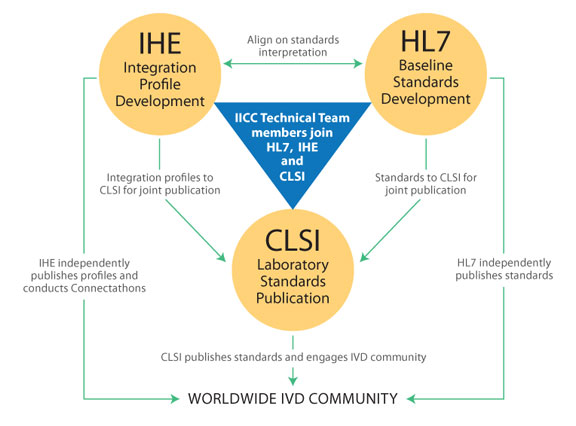
Health Level Seven International (HL7) is the standards development organization (SDO) for IICC. In March 2010, IICC announced the selection of HL7 2.x as the baseline standard for next generation laboratory connectivity. The Orders & Observations Working Group within HL7 has responsibility for the IICC initiative, and IICC Technical Team members are members of this workgroup. HL7’s role within the IICC initiative encompasses the following:
Integrating the Healthcare Enterprise (IHE) is the integration profile developer for the IICC initiative. The IHE Laboratory Technical Committee, which includes IICC Technical Team members, has the following roles and responsibilities:
Clinical and Laboratory Standards Institute (CLSI) is the laboratory standards publisher for the IICC initiative. Within CLSI, the Consensus Committee on Automation & Informatics is responsible for the IICC initiative. Its responsibilities:
A new CLSI document development subcommittee, which will include IICC Technical Team members, will be responsible for publishing the standard.